
An expert in drug discovery, design and development, Saïd Sebti, Ph.D., holds 80 patents and several technology licenses for his scientific inventions. He has co-developed two drugs that have reached clinical trials, published more than 300 manuscripts in peer-reviewed academic journals, and has been continuously grant funded by the National Cancer Institute since 1989. Photo: John Wallace, VCU Massey Cancer Center
Signal Disruptions
Editor’s Note: This is an excerpt of a feature that appeared in the summer 2021 issue of NEXT magazine. Our online version includes the full article and more stories about innovative research happening on the MCV Campus.
***
Saïd Sebti can trace his interest in combatting some of the most vicious cancers to a sleepless summer night in his teens. He vividly remembers visiting a friend in Rabat, Morocco, and being kept awake most of the night by the agonizing sounds and screams of pain he could hear from his friend’s next-door neighbor, a woman he would learn the next morning was suffering from cancer. As a teen, he wondered why this woman could not simply go to a doctor to receive medication and relief from her torment. That moment stuck with a 14-year-old Sebti and has been a strong undercurrent in his drive to discover potential solutions to cancer. He earned his doctorate in the U.S. and has steadily worked to shine new light on what causes various cancers and to develop treatments that might work against those causes.
 Today, Saïd Sebti, Ph.D., is the associate director for basic research at VCU Massey Cancer Center, the Lacy Family Chair in Cancer Research, and a professor of pharmacology and toxicology in the VCU School of Medicine. His lab focuses on understanding the mechanisms by which normal body cells turn cancerous. Cancer develops when mutations occur in some of our genes, which cause aberrant signals that trigger uncontrolled cellular division, growth and metastasis. Dr. Sebti hopes to better understand the aberrant signals that cause cancer and, armed with that knowledge, his team seeks to find novel anti-cancer drugs to disrupt the signals that cause some cells to run amok and wreak havoc on the body.
Today, Saïd Sebti, Ph.D., is the associate director for basic research at VCU Massey Cancer Center, the Lacy Family Chair in Cancer Research, and a professor of pharmacology and toxicology in the VCU School of Medicine. His lab focuses on understanding the mechanisms by which normal body cells turn cancerous. Cancer develops when mutations occur in some of our genes, which cause aberrant signals that trigger uncontrolled cellular division, growth and metastasis. Dr. Sebti hopes to better understand the aberrant signals that cause cancer and, armed with that knowledge, his team seeks to find novel anti-cancer drugs to disrupt the signals that cause some cells to run amok and wreak havoc on the body.
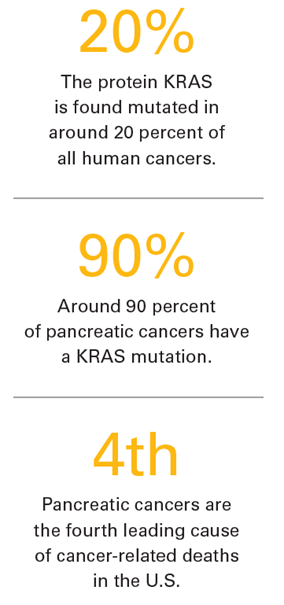
Dr. Sebti’s lab has a special interest in a cancer-causing protein called KRAS that is found mutated in about 20% of all human cancers and which activates many of the aberrant signals of cancer. The mutated KRAS protein is frequently involved in the development of some of the deadliest cancers such as lung, colon and pancreatic. Understanding how mutant KRAS functions is the first step, Dr. Sebti said, to discovering potential ways to interfere with cell processes and signals and hopefully stopping the unfettered growth of cancers.
Prior to his arrival on the MCV Campus, he made major contributions to the understanding of mechanisms by which KRAS proteins cause cancer and identified several drugs that interfere with these processes, particularly in pancreatic cancers. Slightly more than half of pancreatic cancers go undetected until the mutated cells have spread to other parts of the body, where the uncontrolled growth begins to cause more pronounced symptoms. The five-year survival rate for patients is around 10% according to the Centers for Disease Control and Prevention. Cancers of the pancreas are the fourth leading cause of cancer-related deaths in the U.S., and around 90% of pancreatic cancers have a KRAS mutation, which causes more aggressive tumor growth and resistance to chemotherapy and other targeted therapies. The NCI has placed a priority on solutions that target this cancer-causing protein and the development of therapies specific to inhibiting it.
“At present, there are no FDA-approved drugs to directly target mutant KRAS-driven human cancers, and novel drugs are urgently needed for the large number of afflicted patients,” said Dr. Sebti, a member of the Developmental Therapeutics research program at Massey. “We developed several small molecules that overcome the major hurdle of mutant KRAS-dependent resistance that can help with thwarting growth in primary and metastatic tumor samples.”
Dr. Sebti’s lab and collaborators at Moffit and other institutions developed drugs that inhibit the cancer-causing activity of KRAS. The drugs proved effective when applied to human pancreatic, lung and colon tumors in early research. They also prevented tumor growth in mutant KRAS tumor samples from pancreatic cancer patients, some of which were resistant to chemotherapy and radiation therapy.
Since arriving at Massey, in addition to molecular and cellular biologists, Sebti is expanding his research team to include structural biologists and computational biologists, taking a multidisciplinary approach to understanding why some tumors are addicted to mutant KRAS, when others are not. Recently, the Sebti lab discovered a genomic signature that may enable scientists and physicians to predict which patient tumors may be KRAS-addicted and which drugs these tumors may respond to. The MCV Campus work using computational biology has enabled his lab to analyze hundreds of individual cancers in an attempt to understand why some tumors are KRAS-addicted and others are not. These extensive efforts paid off, and what his lab discovered here in Richmond is a set of genes that contribute to the hallmark of KRAS addiction in tumors.
“What is exciting about this is that we’ve identified a genomic signature that could tell us which tumors are, and which are not, KRAS-addicted,” Dr. Sebti said. “If validated in patients, the consequences could be huge — we would have a more refined understanding of what factors to potentially attack and how to pursue therapy development for these cancers.”
Establishing a genomic signature for KRAS addiction in tumors allows clinicians to better understand which patients will respond better to certain treatments, depending on whether their tumor is addicted to KRAS. This gene signature can predict whether patients may have more aggressive forms and poorer outcomes, but this signature also provides the next building block for hope, making it easier to develop drugs that may work best for certain types of cancers.
If you would like to help strengthen the work of Dr. Sebti and Massey researchers working to develop novel therapies to treat cancer, please consider reaching out to the Massey Development Office at 804-828-1450.


